Research
We are laying the foundation for a new field of research informally referred to as “angstrom technology.” We are building biomimetic materials to support a versatile biomimetic toolbox.
Our immediate goal is to engineer an array of intelligent materials spanning scales from angstrom to macro. With this aim in mind, we focus our efforts on
- Exploring complex and dynamic self-assembly using DNA, RNA and proteins or peptides as programmable molecules.
- Using biomolecular self-assembly for the finest positional control of molecules for the construction of interactive spatial networks of chemical and biochemical species.
- Developing multicomponent and multifunctional nanoparticle and nanowire materials for biosensing, bioimaging and energy applications.
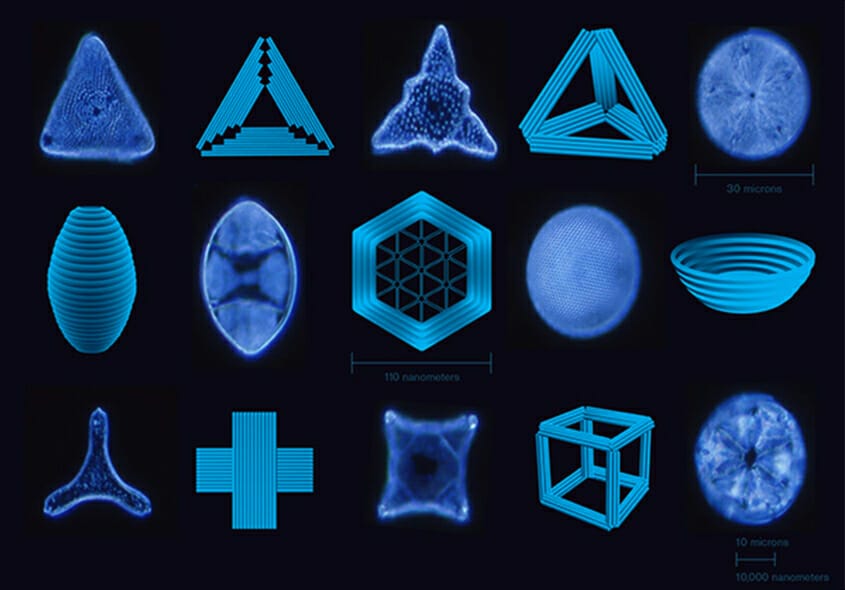
What makes us unique is our capabilities to
- Bring together traditionally separate fields of protein, DNA and RNA nanotechnology.
- Integrate chemistries and materials into applications beyond the limits of biology.
- Combine different types of structural, functional and dynamic components.
- Create far-from-equilibrium systems and exploit their reconfigurability.
- Build systems that can be integrated into human-centered technology with electronic or optical inputs and outputs.
Our areas of investigation are described in more detail below.
Structural DNA nanotechnology
We use DNA as a programmable nanoscale building material to construct designer nanoarchitectures with increasing complexity in 2D and 3D spaces. We are interested in hierarchical engineering of sophisticated nanomachines.

Self-assembling peptide and protein hybrid materials
In addition to DNA, we incorporate proteins and peptides into our nanomaterial assemblies, either exclusively or through hybrid molecules with oligonucleotides. We are particularly interested in systems that combine multiple molecular types to create final assemblies with the advantages of both biomolecules. We aim to create artificial delivery systems (such as synthetic viruses), antibody mimics, nanomachines, novel catalysts and biomaterials.
Bioinspired material
Nature has evolved delicate cellular machineries for all living forms. In biological systems, an ensemble of molecular machines (such as enzyme, cofactor, chaperon, protein motors, etc.) work together and give rise to their cellular functions. Some of the interaction between these functional components is confined by natural scaffold (scaffold protein, for example) and can be tuned dynamically in response to foreign stimulations.
Inspired by this, we are working to organize biomolecules onto deliberately designed DNA scaffold to study distance and spatial-dependent interactions between various biomolecules and ligands. Our goal is to construct self-assembled, dynamic and functional biomolecular networks for therapeutic applications.
Computer modeling of DNA and RNA nanotechnology
We develop new computational and design tools (oxDNA and oxView) to simulate nucleic acid nanotechnology. The tools bring part of the design process in silico and allow us to rationalize experimental results and probe properties of systems at resolution that is not possible in experiments.
Machine learning for RNA and DNA sequences
We employ models of machine learning and data analysis to study sequence and structural motifs in RNA and DNA molecules that are indicative of their function or properties. To do this, we combine molecular simulation with bioinformatics.
Nanophotonics and nanoelectronics
Metallic and semiconducting nanoparticles possess unique optoelectrical properties. The organization power of DNA makes it possible to position discrete numbers of nanoparticles in 2D and 3D with nanometer precision, which offers an ideal platform to study the collective properties of nanoparticles. Emerging photonic properties are expected from designer nanoparticle architectures for photonic device applications. DNA directed assembly of nanoparticles, in conjunction with surface patterning techniques, provides a promising route for massively parallel fabrication of nanodevices.
Single molecule nanoarrays
The programmability and biocompatibility are the most unique and treasurable properties that self-assembling DNA nanoarrays offer. We seek to build single-molecule nanoarray-based platforms for the multiplexed detection of nucleic acid, protein, and other biologically relevant molecules at the single cell level. Probes with specific molecular binding abilities (such as aptamers and antibodies) are incorporated in the nanoarray for protein and nucleic acid profiling.
Biosensing
We are developing a liquid-biopsy method for low-cost cancer diagnostics. Inspired by signal-transducing membrane proteins, we design and synthesize DNA devices that sense biomarkers inside intact exosomes. Currently, almost all exosome-based diagnostics require breaking up the compartments. By keeping the exosomes intact, our innovative approach is expected to advance biomedical research by providing rich insights into the distribution of biomarkers at the single-exosome level.
Inorganic nanomaterial synthesis and bioconjugation
Many of the above research projects require the preparation of water-soluble and biomolecule conjugated nanoparticles. We are interested in developing simple and robust methods for creating stable water-soluble nanoparticle-biomolecule conjugates. Such bioconjugated nanoparticles are necessary to build multicomponent and multifunctional hierarchical structures of unique properties.
Self-replication and dynamic molecular assembly
We pursue in vitro and in vivo replications of nucleic acid-based nanostructures for scaling up and evolving functional nanostructures. Another important aspect of our research is to create self-assembling structures that are dynamic to allow the trigger of a cascading molecular event, using either small molecules or biological molecules as logical inputs. We seek to understand collective behaviors of different molecules interacting with each other.
Multifunctional nanostructures interfacing with cellular systems
Our research explores developing geometrically defined and multifunctional nucleic acid nanostructures that target specific organelles of biological cells for potential applications in cancer therapy and drug delivery.
Biomaterials for regenerative medicine
The future of medicine will rely on regeneration of tissue damaged by injury or disease. We are constructing artificial cell scaffolds from proteins, peptides and DNA that can recapitulate the extracellular environment and stimulate healing (either of endogenous cells or injected stem cells). In particular, we focus on materials that can translate between multiple different length scales and dynamic materials that can control the matrix properties in a tunable and multiplexed fashion.
Support the Biodesign Center for Molecular Design and Biomimetics
Using nature’s design rules as an inspiration in advancing biomedical, energy and electronics innovation.